Abstract
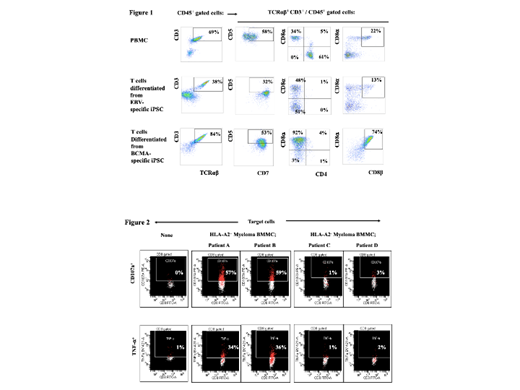
T cell regenerative medicine represents an emerging immunotherapeutic approach using antigen-specific Induced Pluripotent Stem Cells (iPSC) to rejuvenate CD8 + cytotoxic T lymphocytes (CTL). Here we report on an iPSC-derived therapeutic strategy targeting B-Cell Maturation Antigen (BCMA) against multiple myeloma (MM) via establishment of antigen-specific iPSC, followed by differentiation into highly functional BCMA-specific CD8 + CTL. The reprogrammed BCMA-specific iPSC displayed normal karyotypes and pluripotency potential as evidenced by expression of stem cell markers (SSEA-4, TRA1-60) and alkaline phosphatase, along with differentiation into three germ layers (Ectoderm, Mesoderm, Endoderm). During embryoid body formation, BCMA-specific iPSC further polarized into the mesoderm germ layer, evidenced by the activation of SNAI2, TBX3, PLVAP, HAND1 and CDX2 transcriptional regulators. Next, the BCMA-specific iPSC clones committed to CD8 + T cell differentiation were characterized by analyzing their hematopoietic progenitor cells (HPC; CD34 + CD43 +/CD14 - CD235a -) for specific transcriptional regulation. RNAseq analyses indicated a low variability and similar profiles of gene transcription within the iPSC clones committed to CD8 + CTL compared to increased transcriptional variability within iPSC clones committed to different cell types. The unique transcriptional profiles of the iPSC committed to CD8 + T cells included upregulation of transcriptional regulators controlling CD4/CD8 T cell differentiation ratio, memory CTL formation, NF-kappa-B/JNK pathway activation, and cytokine transporter/cytotoxic mediator development, as well as downregulation of regulators controlling B and T cell interactions, CD4 + Th cells, and inhibitory receptor development. Specifically, a major regulatory shift, indicated by upregulation of specific genes involved in immune function, was detected in HPC from the iPSC committed to CD8 + T cells. BCMA-specific T cells differentiated from the iPSC were characterized as displaying mature CTL phenotypes including high expression of CD3, CD8a, CD8b, TCRab, CD7 along with no CD4 expression (Fig. 1). In addition, the final BCMA iPSC-T cells were predominantly CD45RO + memory cells (central memory and effector memory cells) expressing high level of T cell activation (CD38, CD69) and costimulatory (CD28) molecules. Importantly, these BCMA iPSC-T cells lacked immune checkpoints (CTLA4, PD1, LAG3, Tim3) expression and regulatory T cells induction, distinct from other antigen-stimulated T cells. The rejuvenated BCMA iPSC-T cells demonstrated a high proliferative (1,000 folds increase) during the differentiation process as well as poly-functional anti-tumor activities and Th1 cytokine (IFN-g, IL-2, TNF-a) production triggered in response to MM patients' cells in HLA-A2-restricted manner (Fig. 2). Furthermore, the immune responses induced by these BCMA iPSC-T cells were specific to the parent heteroclitic BCMA 72-80 (YLMFLLRKI) peptide used to reprogram and establish the antigen-specific iPSC. Evaluation of 88 single cell Tetramer + CTL from the BCMA iPSC-T cells revealed a clonotype of unique T cell receptor (TCRa, TCRb) sequence. The BCMA-specific iPSC clones maintained their specific differentiation potential into the antigen-specific CD8 + memory T cells, following multiple subcloning in long-term cultures under feeder-free conditions or post-thaw after long-term (18 months) cryopreservation at -140 oC, which provides additional benefits to treat patients in a continuous manner. Taken together, rejuvenated CD8 + CTL differentiated from BCMA-specific iPSC were highly functional with significant (*p < 0.05) levels of anti-MM activities including proliferation, cytotoxic activity and Th-1 cytokine production. Therefore, the antigen-specific iPSC reprogramming and T cells rejuvenation process can provide an effective and long-term source of antigen-specific memory CTL lacking immune checkpoints and suppressors for clinical application in adoptive immunotherapy to improve patient outcome in MM.
Munshi: Amgen: Consultancy; Karyopharm: Consultancy; Takeda: Consultancy; Adaptive Biotechnology: Consultancy; Bristol-Myers Squibb: Consultancy; Celgene: Consultancy; Abbvie: Consultancy; Janssen: Consultancy; Legend: Consultancy; Oncopep: Consultancy, Current equity holder in publicly-traded company, Other: scientific founder, Patents & Royalties; Novartis: Consultancy; Pfizer: Consultancy. Ritz: Amgen: Research Funding; Equillium: Research Funding; Kite/Gilead: Research Funding; Avrobio: Membership on an entity's Board of Directors or advisory committees; Akron: Consultancy; Biotech: Consultancy; Blackstone Life Sciences Advisor: Consultancy; Clade Therapeutics, Garuda Therapeutics: Consultancy; Immunitas Therapeutic: Consultancy; LifeVault Bio: Consultancy; Novartis: Consultancy; Rheos Medicines: Consultancy; Talaris Therapeutics: Consultancy; TScan Therapeutics: Consultancy. Anderson: Sanofi-Aventis: Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Millenium-Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Scientific Founder of Oncopep and C4 Therapeutics: Current equity holder in publicly-traded company, Current holder of individual stocks in a privately-held company; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Mana Therapeutics: Membership on an entity's Board of Directors or advisory committees.